Research Misconduct
At Hackensack Meridian Health, we are dedicated to the highest standards of integrity in all our research. Upholding these standards is essential for advancing medical science, ensuring patient safety, and maintaining public trust.
We believe that every member of our network shares the responsibility to ensure that our research is conducted ethically and responsibly. Hackensack Meridian Health is committed to addressing any concerns about research misconduct promptly, thoroughly, and fairly.
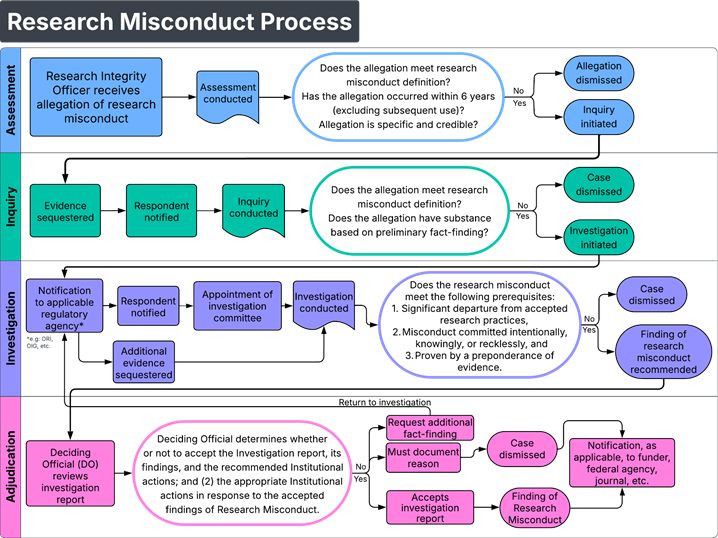
HMH’s Research Compliance division is responsible for overseeing and administering HMH’s Policy on Research Misconduct. The Research Compliance Officer is the designated Research Integrity Officer (RIO) and is a resource for anyone with concerns or questions about possible research misconduct.
HMH defines research misconduct, in accordance with federal regulations, as any fabrication, falsification, or plagiarism in the proposing, performing or reviewing of research or reporting of research results. Research misconduct does not include honest error or differences of opinion.
|
Type of Research Misconduct |
Definition |
|
Fabrication |
The making up of data or results and the recording or reporting thereof. |
|
Falsification |
The manipulation of Research materials, equipment or processes, or the change or omission of data or results such that the Research is not accurately represented in the Research Record. |
|
Plagiarism |
The appropriation of another person’s ideas, processes, results or words without giving appropriate credit. |
If You Have a Concern
- Discuss with a trusted chair, supervisor or other official
- Contact the Research Compliance Officer, Dr. Michelle Benson
- Anonymous reporting via HMH’s ComplyLine
Retaliation of any kind, against those who report a concern in good faith is not tolerated by HMH.